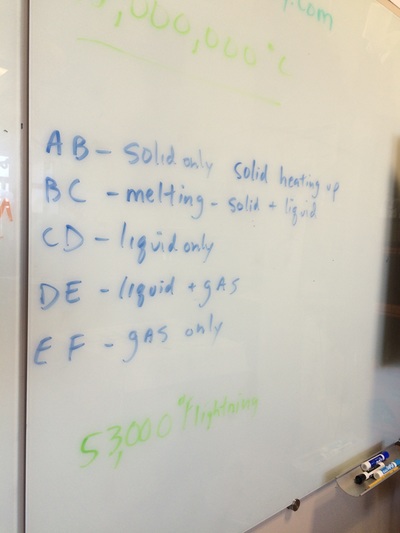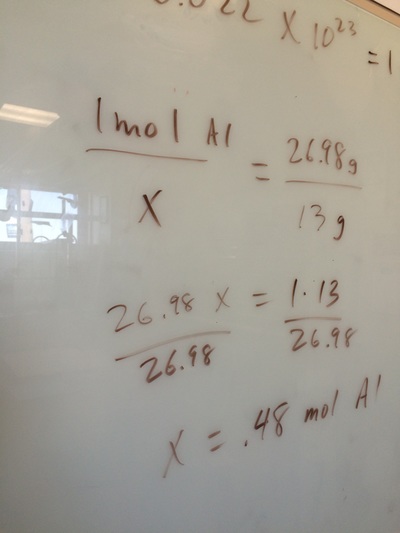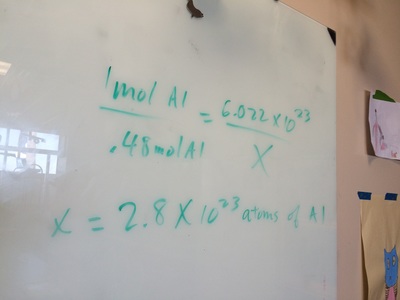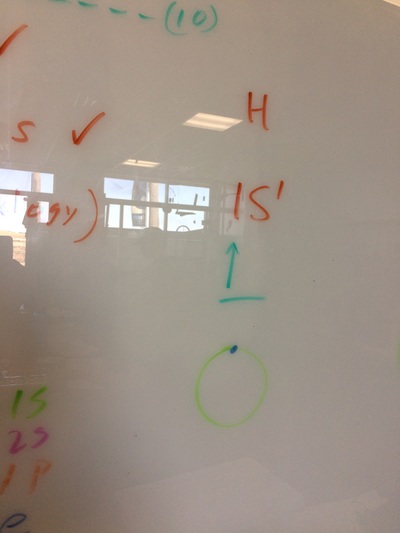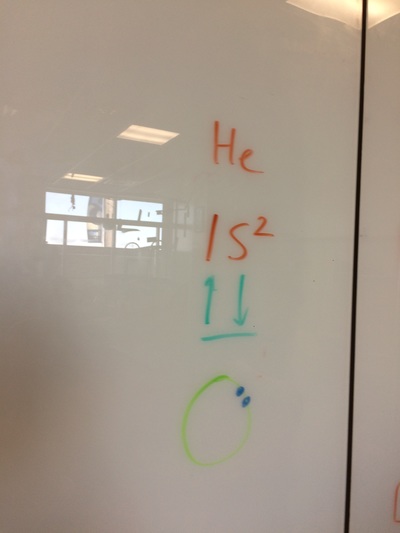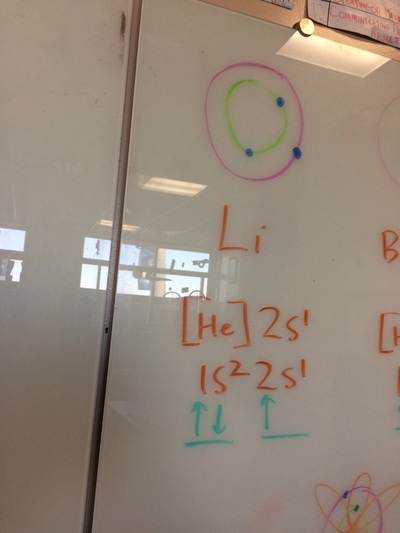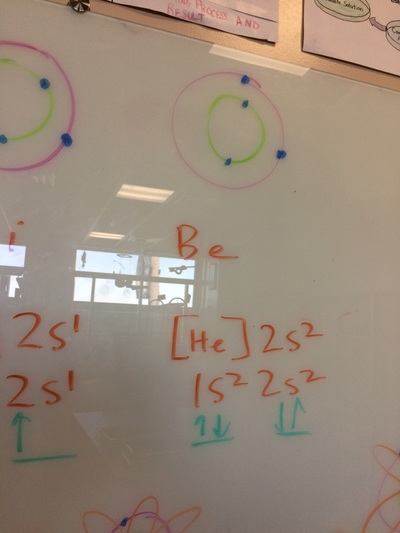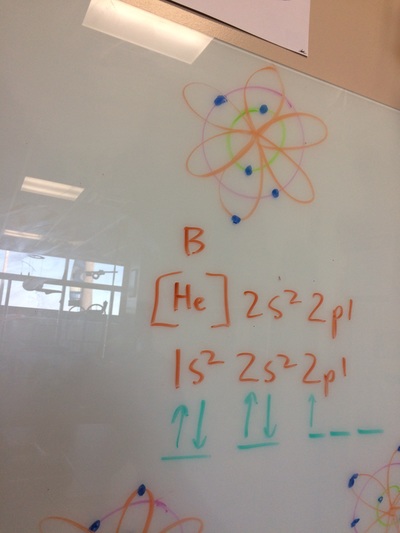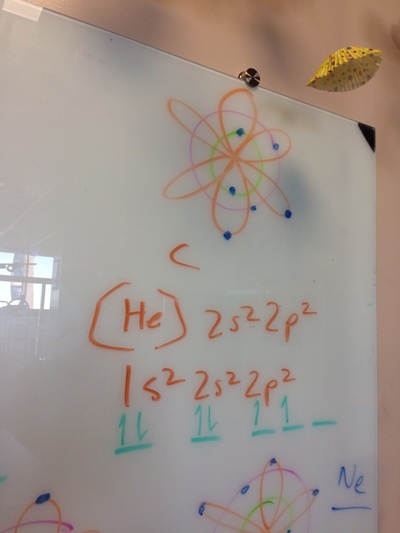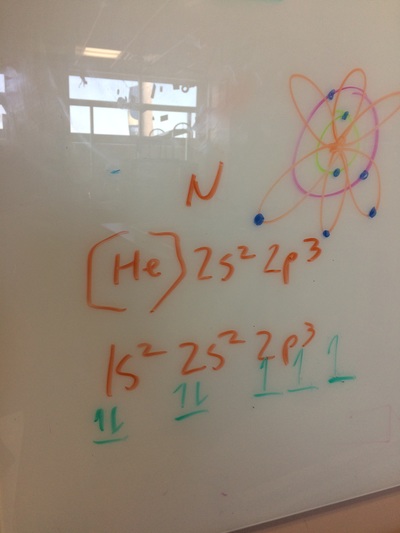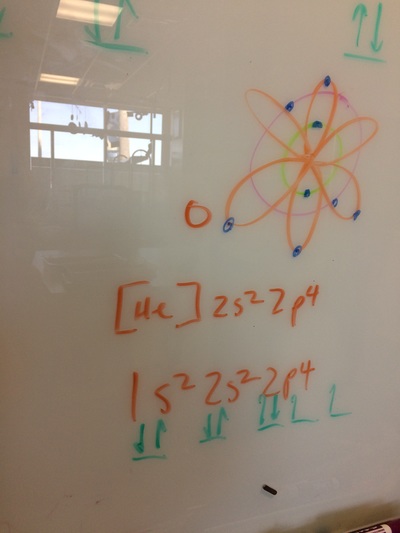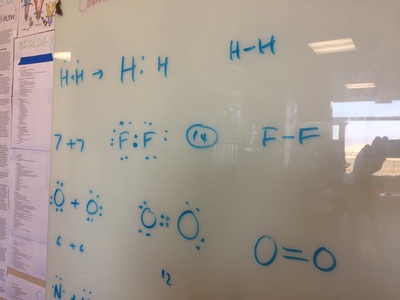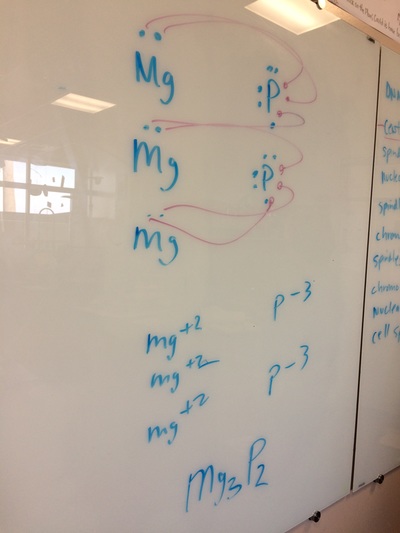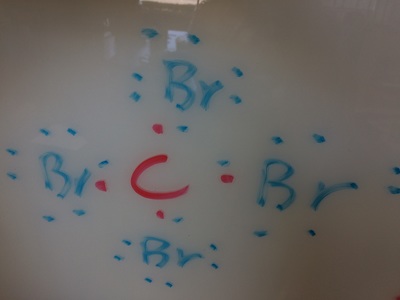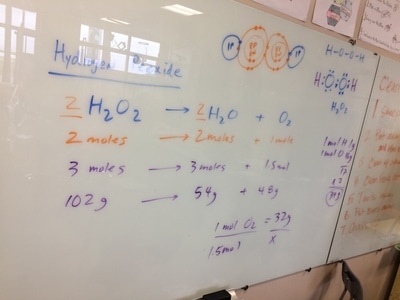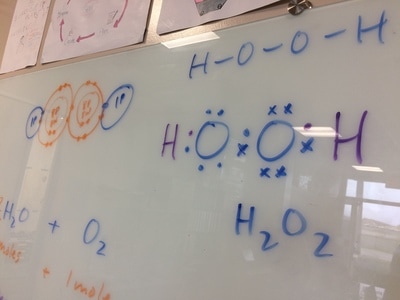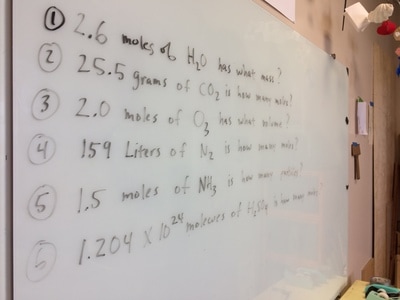Chemistry is taught as a concept oriented course with emphasis on understanding the general laws governing the properties of the elements, their compounds and their reactions. Importance will be placed on understanding how chemical principles and concepts are developed and how these principles can be used to explain phenomena in everyday life. Ample mathematical application is made in all topics. Students will explore chemistry through the study of the following strands: Atomic and Molecular Structure, Chemical Bonds, Conservation of Matter and Stoichiometry, Gases and their Properties, Acids and Bases, Solutions, Chemical Thermodynamics, Reaction Rates, Chemical Equilibrium, Organic Chemistry and Nuclear Processes. This course is recommended for students who are university bound (meets A-G requirements). Completion of Chemistry A/B fulfills the physical science graduation requirement. Completion of Biology A/B and concurrent enrollment in Geometry or Algebra 2 is recommended.**Students taking this course for UC credit must follow the guidelines listed on the Course Description Form.
CLICK HERE TO SEE A FULL COURSE OVERVIEW.
CLICK HERE TO SEE A FULL COURSE DESCRIPTION.
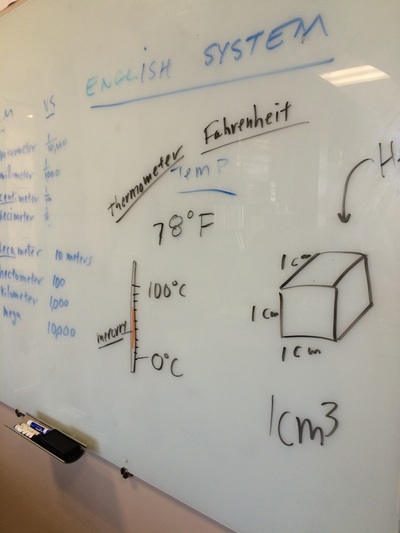
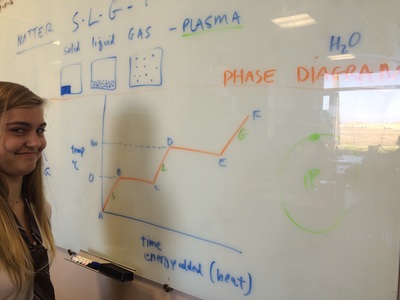
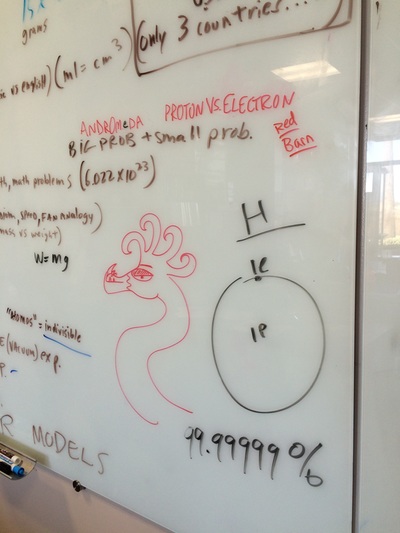
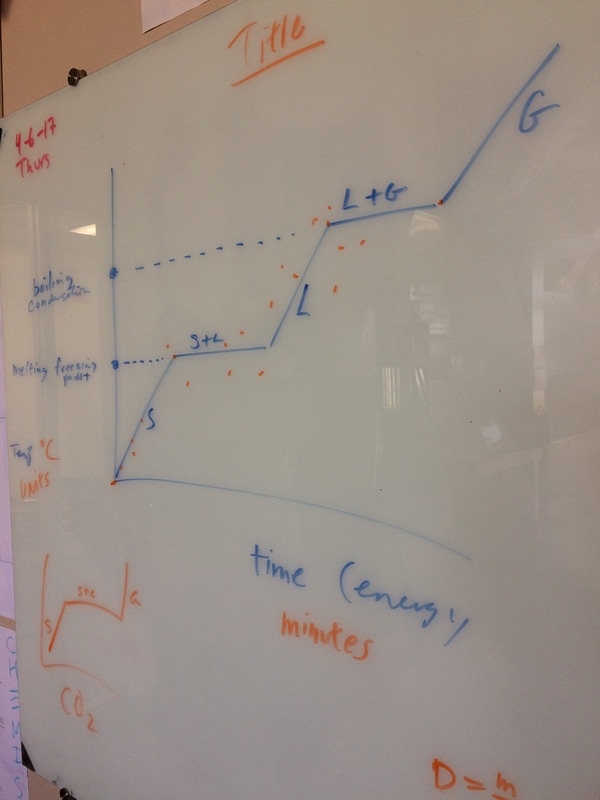
We hit the ground running this year and with three hours a week we can cover a lot of ground! We will do our best to follow the outline in the course description but retain the right to meander whenever student interest or a relevant tangent presents itself! Please find below photo-documentation from our first class where we delved into atoms, energy, volume, measurement, states of matter, temperature, phase diagrams, why the metric system is way cooler than the standard English system, plasma, etc!
Here's a video we watched starring the inimitable Hank Green, thanks Hank!
The World's Smallest Movie:
How It Was Made:
Atoms are REALLY Small:
J.J. Thompson-Cathode Ray Experiment
J.J. Thompson discovered that there are particles smaller than the atom, that these particles were negatively charged, and they flew through a vacuum!! All this way back in 1897! Watch this video to learn more about it!
Ernst Rutherford performed an incredible experiment with gold foil and alpha particles thus discovering that the atom was 99.9999% empty space!
Robert Millikan, in 1909, came up with an incredibly creative experiment that enabled him the measure the charge on a single electron: 1.602x10-19 Coulombs!
More Hank Green please!
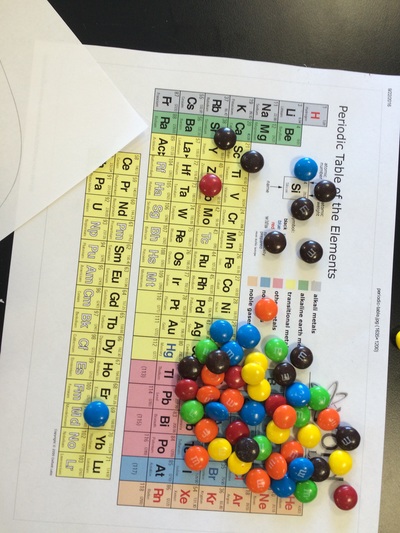
Bohr Models, atomic structure, protons, electrons, neutrons, and M&M's.
An incredible song about the sun, nuclear fusion, from an incredible band, They Might be Giants!
Another incredible song about the elements by the inimitable Tom Lehrer!
The periodic Table of Videos!!! YES, it is true! One video for almost all of the elements! Grab some popcorn and enjoy the show!!
The alkali metals react with water:
Salt (NaCl) Formation:
Introduction to the MOLE and AVOGADRO'S NUMBER.
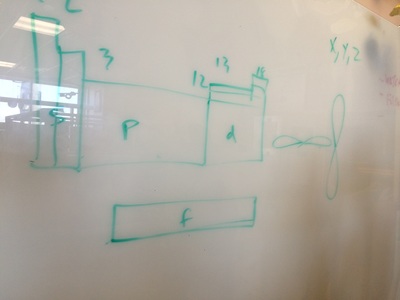
Exploring the shapes and spatial relations of electron orbitals.
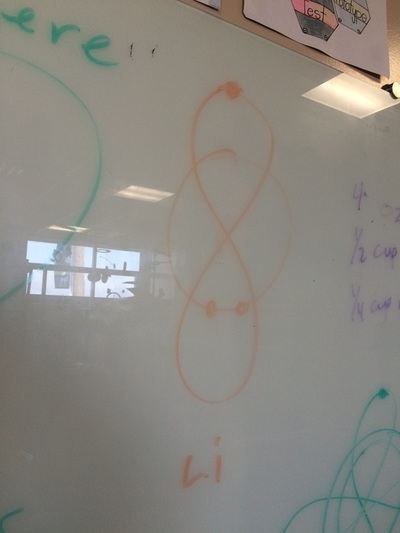
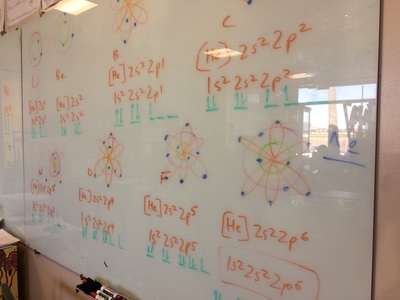
ELECTRON CONFIGURATIONS, ETC!
IONS, IONIC BONDING, ETC!
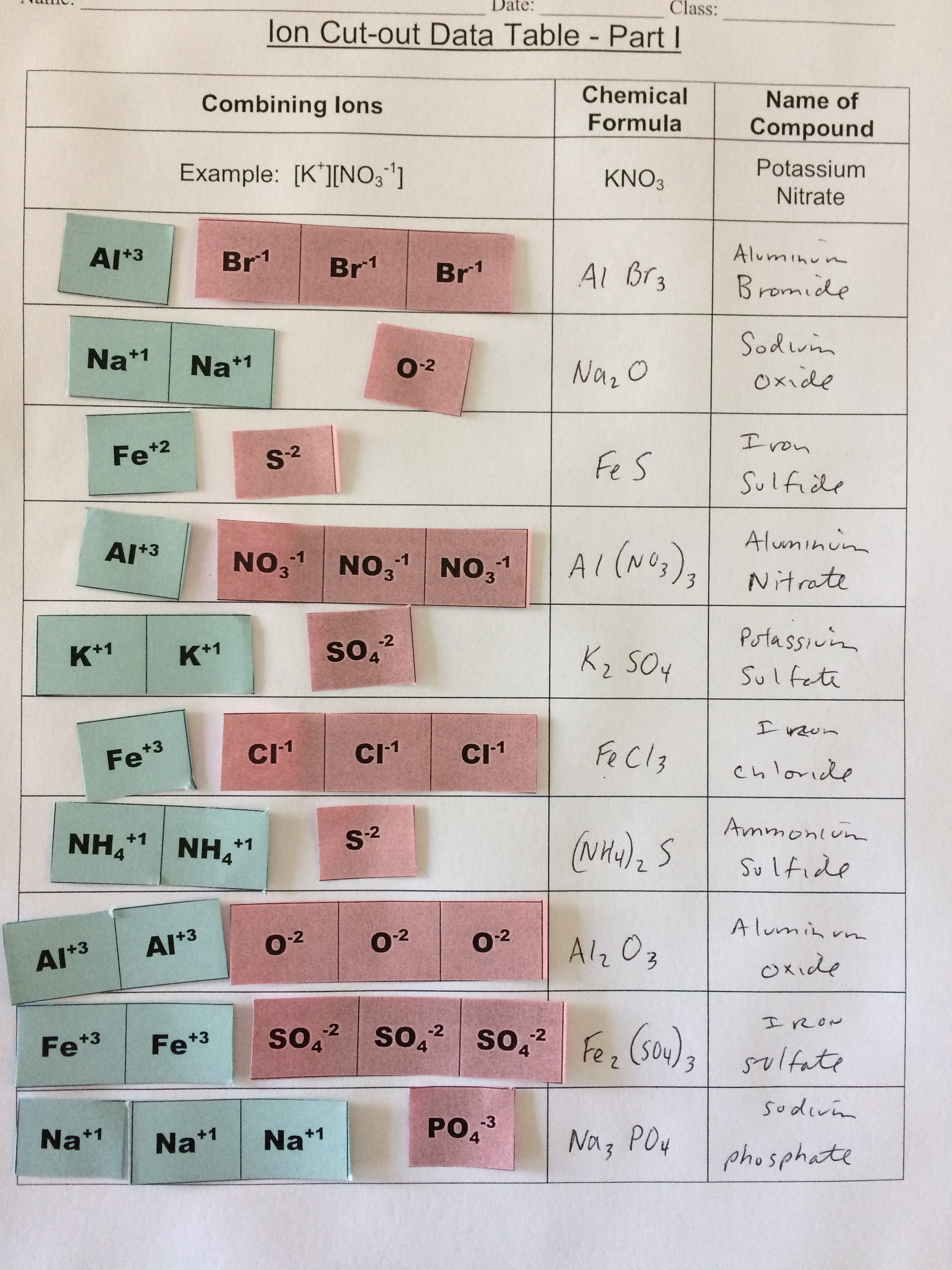
Today, we did a variety of activities to learn about ions, polyatomic ions, ionic bonding, naming ionic compounds, and the crystal lattice. It was great!
Click here to see an ion bonding cutout activity (shown in the photo below!)
Click here to see an ion bonding cutout activity (shown in the photo below!)
BUILDING A CRYSTAL LATTICE!
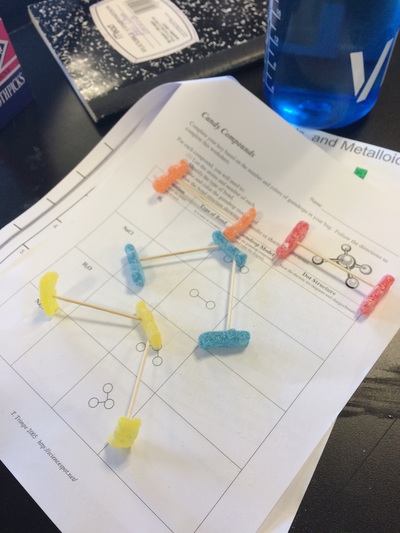
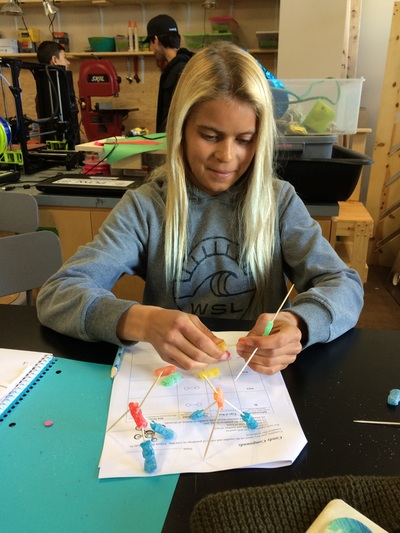
Electronegativity, Ionic, Polar Covalent, Covalent Bonds, and Sour Patch Kids..... all in a days work!
Click here to see part of our lab activities for the day which includes making molecule models and determining the electronegativty of their bonds.
types of equations, balancing equations, molar mass, moles, Lewis diagrams, Bohr models,etc!
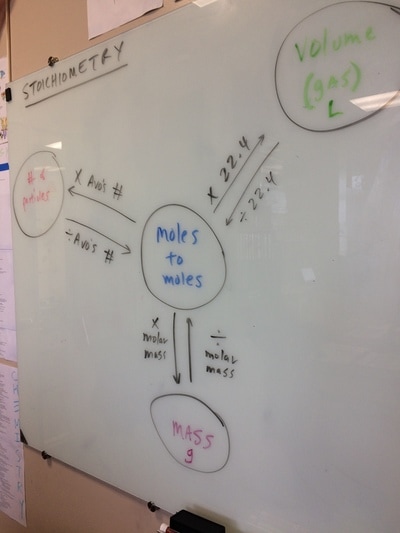
STOICHIOMETRY
FERMENTATION LAB
brewing Ginger Beer and Kombucha!
click here for ginger beer
and here for SCOBY!
GAS LAW LAB
BOYLE'S LAW, CHARLE'S LAW, GAY-LUSSAC'S LAW,
IDEAL GAS LAW, COMBINED GAS LAW
ACIDS AND BASES
RED CABBAGE pH INDICATOR LAB
THERMODYNAMICS
for a Q=mcat introductory worksheet click here!
FERMENTATION LAB- KOMBUCHA!
CALORIMETRY LAB
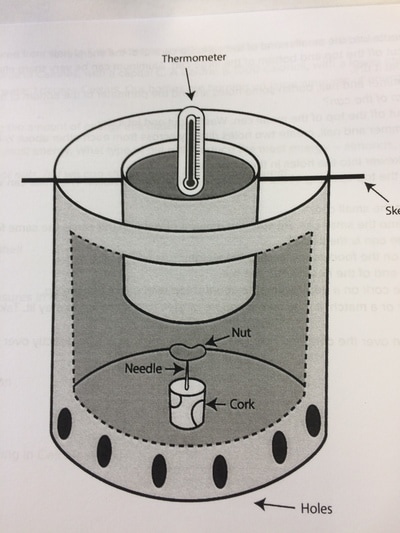
PHASE CHANGE/STATES OF MATTER LABS
Click here to see a phase change lab.
Click here to see a phase diagram worksheet.
Le Chatelier's Principle
Click HERE to check out a worksheet on this principle!